GMP Leukopaks Engineered for Clinical Excellence
HIGH-YIELD, HIGH-QUALITY STARTING MATERIALS TRUSTED IN CELL AND GENE THERAPY TRIALS AND MANUFACTURING.
GMP-compliant leukopaks for critical applications in cell and gene therapy, collected, processed, and delivered with unmatched precision and regulatory assurance.

GMP Fresh Clinical Leukopaks
CGT Clinical Leukopaks are collected from IRB-approved consented donors or approved patients at 3 coast-to-coast FDA registered collection centers by qualified and trained staff using the Terumo Spectra Optia® cMNC collection protocol. Donor eligibility is determined following the AABB approved Donor History Questionnaire and in compliance with FDA 21 CFR subpart C infectious disease testing and medical history. CGT collection sites are FDA registered for HCT/P and our laboratories are CLIA registered. Standard infectious disease testing is performed by a qualified provider using FDA-licensed test methods. Collections are shipped fresh for same day, overnight or international delivery and documentation provided is customizable and compliant with GMP. CGT Quality Assurance reviews each batch for accuracy and release
GMP Cryopreserved Clinical Leukopaks
For workflows requiring greater flexibility, our GMP Cryopreserved Clinical Leukopaks are the answer to your needs. With the ability to screen from a large donor pool, our cell-rich leukopaks are collected from I-consented donors at our FDA-registered Clinics. Our leukopaks are isolated in a GMP ISO rated clean room following optimized protocols on the day of collection to ensure the highest quality.
GMP Clinical Whole Blood
CGT collects Clinical Whole Blood at all collection sites by qualified and trained staff under an approved Batch Record using CPD or ACD-A anticoagulant bags. Donor eligibility is determined following the AABB approved Donor History Questionnaire and in compliance with FDA 21 CFR subpart C infectious disease testing and medical history. CGT collections are performed on-site at our CLIA registered and HCT/P FDA registered cell collection centers. A qualified provider performs standard infectious disease testing using FDA-licensed testing methods. Documentation can be customized to meet project needs and GMP compliance. CGT Quality Assurance reviews each batch for accuracy and release before product is shipped fresh for same day, overnight or international delivery.
Key Highlights:
-
-
Donor Screening: Ensuring that donors undergo thorough screening procedures to assess eligibility and minimize the risk of transmitting infectious diseases or other adverse events.
-
Aseptic Collection and Processing: Performing leukapheresis and subsequent processing steps under sterile conditions to prevent contamination of the leukopak.
-
Quality Control: Implementing quality control measures to monitor the integrity, purity, and potency of leukopaks throughout the collection and processing process.
-
Documentation and Recordkeeping: Maintaining detailed records of donor information, collection procedures, and processing protocols to facilitate traceability and accountability.
-
Facility and Equipment Compliance: Ensuring that facilities and equipment used for leukopak collection and processing meet GMP standards and are regularly inspected and maintained.
-
Staff Training and Qualification: Staff Training and Qualification: Comprehensive training and education are provided to all personnel involved in leukopak collection and processing to ensure their competency and full compliance with GMP standards.
-
Leukopaks – Clinical Grade Specification
|
Attribute |
Description |
|
Volume |
150–250 mL |
|
Collection Method |
Apheresis via Spectra Optia® |
|
Anticoagulant Used |
ACD-A |
|
Viability Fresh (Cryopreserved) |
≥97% ≥80% post-thaw recovery |
|
Pathogen Testing |
HIV-1/2, HBV, HCV (CLIA panel); additional tests available upon request FDA Licensed testing compliant with 21 CFR Part 1271 for donor eligibility and performed by CLIA licensed laboratory Full Panel details: ◦HIV I & II Ab + Reflex ◦HIV/HBV/HCV NAT ◦HCV Ab ◦HBV Ab Core + Reflex ◦HBV Ab Surface Ag + Reflex ◦HTLV I & II Ab + Reflex ◦Syphilis Ab ◦West Nile Virus NAT ◦T. Cruzi (Chagas) Ab ◦CMV Total ◦CMV Total with Reflex to IgM (Only performed if CMV total test result is positive) ◦CMV Total with Reflex to IgM and IgG/IgM (Only performed if CMV total test result is positive) ◦Zika NAT* inquire
|
|
Donor Screening |
Comprehensive medical history, serology, and IRB-consented |
|
Customization |
Age, gender, HLA-type, BMI, ethnicity |
Whole Blood – Clinical Grade Specifications
|
Attribute |
Description |
|
Volume |
60–500 mL (customizable collection volumes available) |
|
Collection Method |
Venipuncture using sterile, closed system |
|
Anticoagulant Used |
ACD-A (standard) or CPD |
|
Pathogen Testing |
FDA Licensed testing compliant with 21 CFR Part 1271 for donor eligibility and performed by CLIA licensed laboratory Full Panel details: ◦HIV I & II Ab + Reflex ◦HIV/HBV/HCV NAT ◦HCV Ab ◦HBV Ab Core + Reflex ◦HBV Ab Surface Ag + Reflex ◦HTLV I & II Ab + Reflex ◦Syphilis Ab ◦West Nile Virus NAT ◦T. Cruzi (Chagas) Ab ◦CMV Total ◦CMV Total with Reflex to IgM (Only performed if CMV total test result is positive) ◦CMV Total with Reflex to IgM and IgG/IgM (Only performed if CMV total test result is positive) ◦Zika NAT* inquire |
|
Donor Screening |
Full medical history, IRB-consented, serology, and infectious disease screening |
|
Customization |
Donor selection by age, gender, BMI, HLA-type, ethnicity, and disease state |
-
T-cell expansion and engineering for autologous and allogeneic therapies, including CAR-T, TCR-T, and TIL-based immunotherapies
-
CD34+ hematopoietic stem cell (HSC) enrichment for gene therapy, transplantation, and regenerative medicine applications
-
Natural Killer (NK) cell isolation and expansion for off-the-shelf immunotherapies
-
Gene editing workflows, including CRISPR-based cell modification for early-phase clinical trials
-
iPSC generation from PBMCs for stem cell research and personalized cell therapy
-
Donor-matched material sourcing for autologous and allogeneic clinical trials
-
Immunophenotyping and functional assays to evaluate immune response in clinical research
-
GMP assay development and validation, including flow cytometry, ELISpot, and qPCR methods
-
Biomarker discovery and immune profiling for patient stratification in clinical studies
-
Companion diagnostic development in support of targeted therapeutic products
-
Cell therapy process development and GMP tech transfer, using clinical-grade input materials
-
GMP cryopreservation protocol development to optimize viability and cell recovery
-
Stability testing and quality control workflows for clinical-grade reagents and devices
-
Training and simulation for GMP-compliant cell therapy manufacturing environments
Meet Your Clinical Team





Why Choose CGT Global’s GMP Leukopaks?
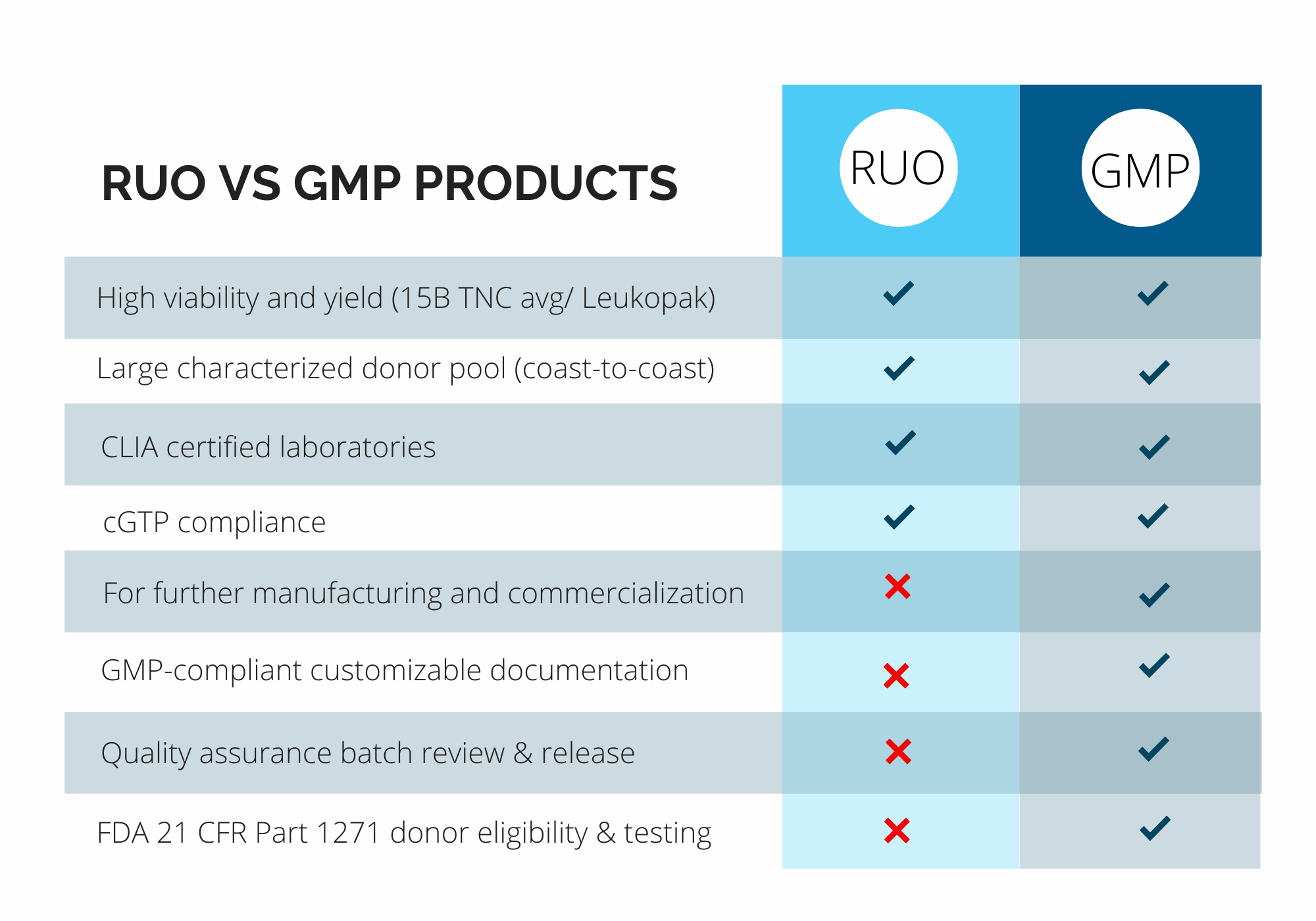
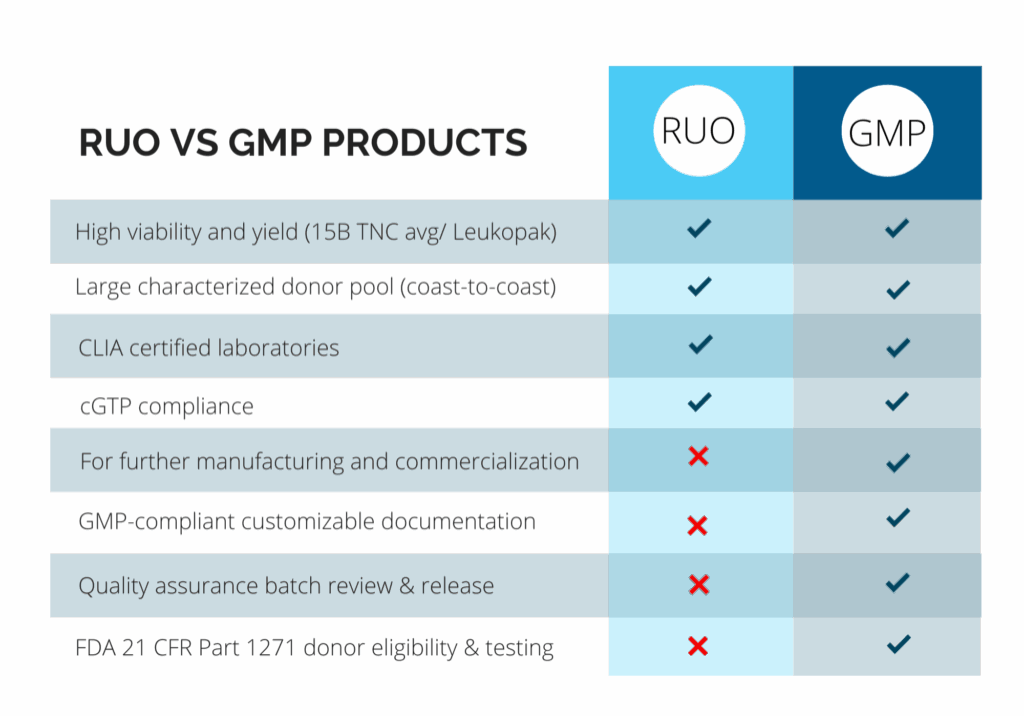
Pre-Clinical (RUO) vs. Clinical (GMP) Products
At CGT Global, we understand that every project is different. That’s why we work closely with each client to determine the specific compliance requirements based on how our materials will be used.
With over a decade of experience as a trusted provider of RUO and GMP-compliant healthy donor materials, we offer flexible, customizable solutions to support researchers and drug developers at every stage of their work.
Partner with CGT Global, where expertise meets flexibility.
Dependable Donors for Reproducible Science
At CGT Global, we recognize that allogeneic therapies, such as CAR-T, demand the right donor, qualified through rigorous screening and compliant with 21 CFR 1271 Subpart C. These donors must meet strict eligibility requirements and often undergo extensive testing before contributing raw material for clinical manufacturing.
To address this critical need, we offer a unique, program-specific donor solution managed by our specialized donor recruitment and collection team. Whether you’re launching a new program or scaling an existing one, our team works closely with you to build a tailored strategy that supports every stage, from donor recruitment through to product fulfillment.
Start Your Program with Confidence
CGT Global will guide you through the startup and ordering process, collaborating with you to deliver a solution that fits your exact needs. Contact us today to schedule an introductory or exploratory evaluation of your program
Common process customizations:
- Location-specific donor collection in geographic proximity to your Manufacturing facility
- Complex donor screening and pre-qualification
- Specific donor recruitment or eligibility criteria
- Additional infectious disease testing (HHV6, 7, 8, Babesia, Toxoplasma, etc.)
- IRB-approved donor Informed Consent Forms (ICF)
- Supplemental Donor History Questionnaires (DHQ)
- Customized Certificate of Analysis (CoA)
- Analytical testing & other donor or collection characterization
- Shipping containers and carriers
This service includes: Leukapheresis, Whole Blood, Cryopreserved PBMCs
- FDA compliant Infectious Disease Testing panels
- Additional Infectious Disease Testing provided by approved vendors
- Donor characterization including HLA, KIR, genomic screening
- Location-based donor recruitment for supply chain optimization
- Dedicated donor management to your clinical program
- Further customization by evaluation
.
FAQ's
-
- Clinical-Grade Materials On Demand
GMP-compliant Leukopaks and whole blood from FDA-registered, CLIA-certified state of the art laboratories, ready for regulatory pathways and high-quality trial consistency. - Speed & Reliability with Coordinated Logistics
Rapid, same-day collection and shipment at the major Biotech hubs in the Massachusetts, Philadelphia and California. — and worldwide shipemnt with our trusted logistics partners. Through our trusted logistics partners, we ensure reliable global delivery of fresh and cryopreserved materials with validated cold-chain systems. Our team proactively manages documentation and compliance to prevent customs delays, ensuring your time-sensitive materials move seamlessly across borders and arrive on schedule. - Advanced Cell Processing for Trial-Ready Materials
Automated isolations via CliniMACS Prodigy® and BioSpherix Xvivo X2®—closed-system, clinical-grade cells custom-prepared to match your protocol. - Regulatory & Documentation Support
Full traceability, IND-ready documentation, IRB support, and donor recall options to streamline compliance and reduce risk. - Scalable Partnership for Every Trial Phase
From preclinical to pivotal trials, we flex with your needs, supporting custom donor projects, specialty isolations, and multi-site studies. - Expert Collaboration, Not Just a Vendor
Dedicated clinical team, responsive service, and over 15 years of experience guiding cell and gene therapy innovators to commercialization.
- Clinical-Grade Materials On Demand
Good Manufacturing Practice (GMP) refers to a set of regulations ensuring that products are consistently produced and controlled according to high-quality standards. It guarantees that the starting materials for clinical trials are safe, effective, and reproducible.
CGT Clinical Leukopaks are collected from IRB-approved consented donors or approved patients at 3 coast-to-coast FDA registered collection centers by qualified and trained staff using the Terumo Spectra Optia® cMNC collection protocol. Donor eligibility is determined following the AABB approved Donor History Questionnaire and in compliance with FDA 21 CFR subpart C infectious disease testing and medical history.
We offer both fresh and cryopreserved GMP-compliant Leukopaks. Our Leukopaks can be customized for specific cell isolation needs, including T-cells, B-cells, NK-cells, and more.
GMP Leukopak is shipped immediately after collection for same-day or next day delivery through our trusted logistics vendors censuring fast and reliable delivery for your projects.
Yes, we provide highly customizable cell isolations from our GMP-compliant Leukopaks. Whether you need specific immune cell types or require a tailored process, our team is here to support your needs.
Our GMP-compliant Leukopaks and Whole Blood are collected under strict sterile protocols. GMP Cryopreserved Leukopaks are tested in accordance with state and Federal requirements for sterility and other pathogen testing.
Tour Our GMP Cell Processing Facility
Step inside CGT Global’s advanced GMP laboratory, where precision meets compliance. Our facility is purpose-built to support clinical-grade cell therapy manufacturing, providing a controlled environment for the processing, expansion, and cryopreservation of critical cell populations.
Cryogenic Storage & Preservation
Our facility houses LN2 cryogenic storage systems, including vapor-phase cryo tanks equipped with automated temperature monitoring and backup power redundancy to maintain sample integrity at all times. All cryopreserved materials undergo validated controlled-rate freezing protocols and are tracked through our 21 CFR Part 11-compliant inventory system.
Closed-System Cell Processing with CliniMACS Prodigy®
We utilize Miltenyi Biotec’s CliniMACS Prodigy® for automated, closed-system cell isolation and enrichment—minimizing contamination risk and supporting GMP-grade reproducibility. For phenotypic analysis of expanded cell populations, we employ MACSQuant® analyzers. Together, these technologies support applications such as CD34+ enrichment, T-cell subset selection, and detailed immunophenotyping.
Integrated QC & Traceability
Samples move through a fully integrated workflow that includes:
-
Real-time viability and phenotyping via flow cytometry
-
Mycoplasma and endotoxin testing
-
Digital chain-of-custody tracking
We welcome collaborators and clients to tour the facility virtually or in person, and witness firsthand the rigor and reliability that define CGT Global’s clinical operations.







